
Three numbers tell the story: 3,000+ units per shift, <1% defects, 99%+ first‑pass yield.
With FDA QMSR now fully effective and EU MDR tightening process validation requirements, manual assembly is no longer just inefficient—it is a regulatory liability.
The global nebulizer market is projected to reach USD 1.67 billion by 2034 (CAGR 6.3%), while the medical‑use segment alone is expected to climb from USD 2.66 billion in 2025 to USD 4.65 billion by 2032, growing at 8.3%.
At the same time, the global medical device assembly automation equipment market is expanding from USD 2.28 billion in 2025 to USD 4.35 billion by 2032 at a CAGR of 9.7%.
Manufacturers who embrace automation now will capture market share—while those who hesitate will be squeezed by rising labor costs, quality escapes, and compliance risk.
Table of Contents
Toggle📊 The Math That Changes Everything
| Metric | Manual Line | RuiZhi Nebulizer Assembly Machine |
|---|---|---|
| Operators per shift | 8–10 | 2 |
| Output per shift | 800–1,200 units | 3,000–5,000+ units |
| Defect rate | 3–5% | <1% |
| Material waste | Baseline | 10–15% reduction |
| Payback period | — | 12–24 months |
The comparison is not subtle. A manual line produces roughly 800–1,200 units per day with 8–10 operators. Our fully automatic nebulizer assembly machine, operating with just two skilled technicians, delivers 3,000 to 5,000+ units per shift—three to five times the output—while cutting direct labor by 70–80% and recovering investment within 12 to 24 months.
🎯 Precision That Regulators Expect

Nebulizer performance depends on tight tolerances: nozzle alignment (±0.1 mm matters), reservoir seal integrity, and proper electronic connections for mesh‑driven or vibrating‑membrane circuits. Manual lines cannot maintain this consistency across shifts, operator fatigue, and product variations.
Our machine achieves ±0.15 mm positioning accuracy using high‑response servo motors and smart sensors—and can be configured down to ±0.02 mm with real‑time force control. Key processes—nozzle installation, sealing ring placement, reservoir assembly, PCBA docking, and shell fitting—are fully automated and continuously monitored in a closed‑loop adaptive system.
🔍 Adaptive In‑Line Quality Control: The Compliance Advantage
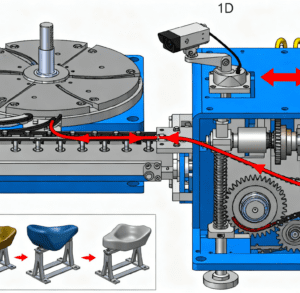
Traditional end‑of‑line sampling catches defects after material and labor are already invested. Our system integrates adaptive quality control at every station, performing automatic in‑line tests: air leak tests, electrical continuity checks, and assembly force verification. The online pass rate consistently exceeds 99%.
For manufacturers subject to ISO 13485, FDA QMSR, and CE audits, this closed‑loop control provides an audit‑ready digital trail that manual assembly cannot offer. Every unit is traceable, every parameter recorded—not because regulation demands it, but because smart manufacturing makes it automatic.
🔧 One Machine, Multiple Models
Jet nebulizers, mesh nebulizers, ultrasonic models—each has its own structure and assembly sequence. Our modular tooling and quick‑change fixture systems let you switch between different SKUs in minutes, not hours. Customizable to fit your exact product specifications, with minimal downtime.
🏭 Built for 24/7 Production
Since 2014, RuiZhi has delivered custom, non‑standard automation solutions to the global medical device industry. Our equipment is engineered for uninterrupted 24‑hour production, featuring robust frames, premium components, and intelligent fault self‑diagnosis. ISO 13485, CE, and FDA compliance are built in, not bolted on.
📈 Why 2026? The Perfect Timing

Three industry tailwinds make 2026 the ideal year to automate your nebulizer assembly:
-
Regulatory convergence: FDA QMSR (effective February 2026) aligns with ISO 13485, emphasizing risk‑based process control and electronic records. EU MDR enforcement is fully underway.
-
AI‑driven quality inspection has moved from pilot to production, enabling real‑time defect detection with greater accuracy than manual inspection.
-
Modular, adaptive machines are revolutionizing medical device manufacturing, allowing one line to handle multiple product variants with minimal changeover time.
#NebulizerAssembly #MedicalDeviceManufacturing #Industry40 #SmartFactory #FDAQMSR




















